Aquatic Ecosystems
Limnology
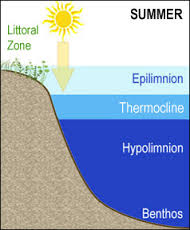
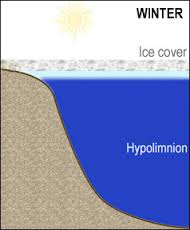
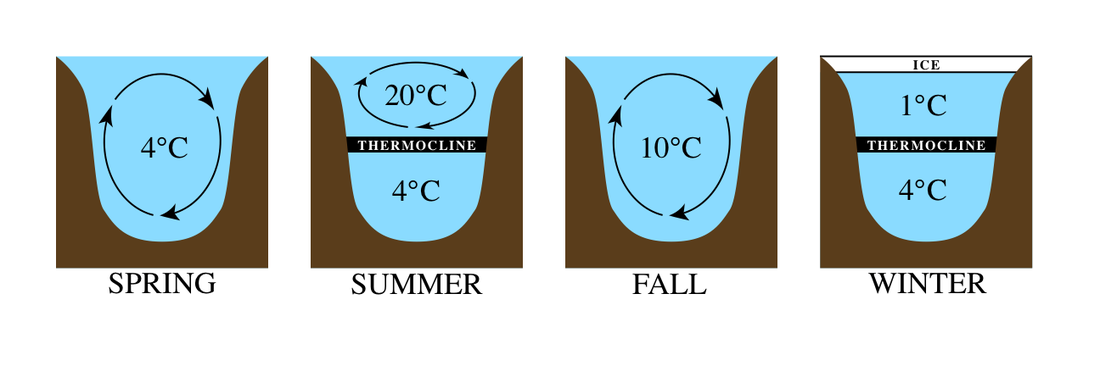
Hyplimnion-the dense, bottom layer of water in a thermally-stratified lake. It is the layer that lies below the thermocline. Typically the hypolimnion is the coldest layer of a lake in summer, and the warmest layer during winter.
Epilimnion- the upper layer of water in a body of water. It is the warmest in the summer and the coldest during the winter.
Thermocline- also known as metalimnion- a layer of water in an ocean or certain lakes, where the temperature gradient is greater than that of the warmer layer above and the colder layer below.
Epilimnion- the upper layer of water in a body of water. It is the warmest in the summer and the coldest during the winter.
Thermocline- also known as metalimnion- a layer of water in an ocean or certain lakes, where the temperature gradient is greater than that of the warmer layer above and the colder layer below.
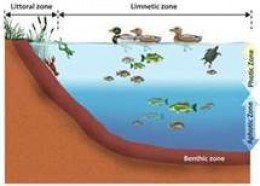
Lentic VS. Lotic
|
Lentic- of or relating to, or inhabiting still water.
What classifies the lentic system?
|
Lotic- of or relating to, or inhabiting moving water.
What classifies the lotic system?
|
Oligotrophic VS. Eutrophic
|
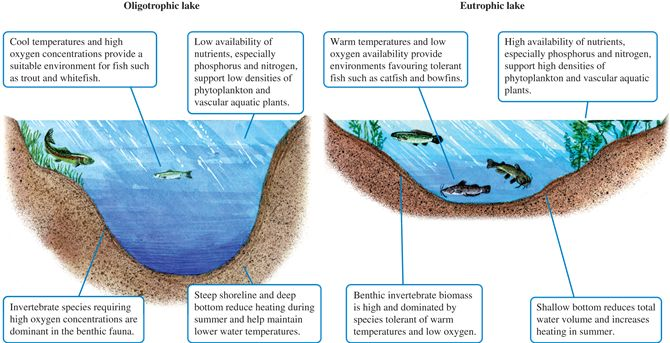
Oligotrophic- lacking in plants and nutrients and having a large amount of dissolved oxygen throughout.
What classifies an oligotrophic zone?
|
Eutrophic- rich in nutrients and so supporting a dense plant population, the decomposition which kills animal life by depriving it of oxygen.
What classifies a eutrophic zone?
|
Is one better than the other? Can they switch between the two? How?
Technically one is not better than the other. Oligotrophic tends to be the one that people rag on all the time because it does not have a plethora of nutrients such as that of the eutrophic zone. But each zone has organisms and plant life that has adapted to its conditions of living, so they are both balanced in their own way. Yes, it is possible to switch between the two zones. If one species or plant life were to invade or take over the environment there, then the system would get out of balance and change to lesser nutrients and so a eutrophic would become an oligotrophic zone. And that goes for the opposite, an oligotrophic zone can change to a eutrophic zone by adding more nutrients to the body of water.
Technically one is not better than the other. Oligotrophic tends to be the one that people rag on all the time because it does not have a plethora of nutrients such as that of the eutrophic zone. But each zone has organisms and plant life that has adapted to its conditions of living, so they are both balanced in their own way. Yes, it is possible to switch between the two zones. If one species or plant life were to invade or take over the environment there, then the system would get out of balance and change to lesser nutrients and so a eutrophic would become an oligotrophic zone. And that goes for the opposite, an oligotrophic zone can change to a eutrophic zone by adding more nutrients to the body of water.
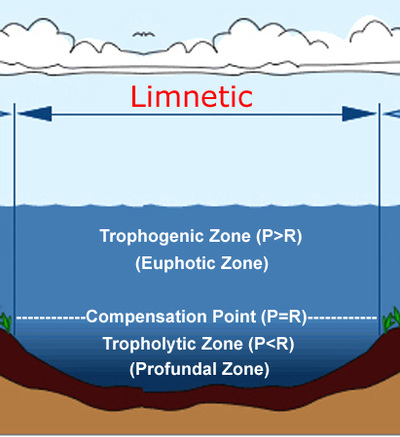
Tropholytic VS. Trophogenic
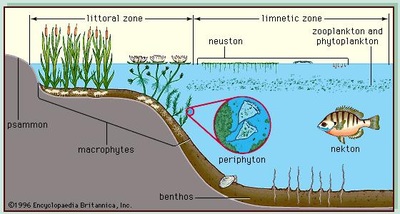
Tropholytic Zone- the region in a lake where light intensity is insufficient to support plants
- Organisms in this zone--> Bottom feeders, crabs, snails, worms
- Organisms in this zone--> trout, catfish, turtles, ducks, frogs
Overturn
- As air temperatures rise in late spring, heat from the sun begins to warm the lake. As the amount of solar radiation absorbed decreases with depth, the lake heats from the surface down. The warm water is less dense than the colder water below resulting in a layer of warm water that floats over the cold water. The layer of warm water at the surface of the lake is called the epilimnion. The cold layer below the epilimnion is called the hypolimnion. These two layers are separated by a layer of water which rapidly changes temperature with depth. This is called the thermocline (or metalimnion). The three distinct layers of water, each with a different temperature or range of temperatures, is an excellent example of thermal stratification within a lake system.
- During the summer the epilimnion will reach a maximum depth and stratification will be maintained for the remainder of the summer. The warm water, abundant sunlight, and nutrients brought up from the lake bottom during spring overturn provide an ideal environment for algae growth within the epilimnion. Algal blooms tend to give the epilimnion a greenish hue. Stratification during the summer acts as a deterrent to complete lake mixing. Wind circulates the surface water, but the warm water of the epilimnion is unable to drive through the cold, dense water of the hypolimnion. As a result, the water is only mixed in the epilimnion.
- As autumn approaches and temperatures decrease, the epilimnion begins to decrease in depth. Eventually the epilimnion gets so shallow that it can no longer be maintained as a separate layer and the lake loses its stratification. Thus, as in the spring, the lake water in the autumn has generally uniform temperatures (about 4°C in late autumn), and wind can once again thoroughly mix the lake water. In addition, surface water, which is in direct contact with the cold air, gets cooled faster than the water below. This cold, dense water sinks and further helps to mix the lake, and once more oxygen and nutrients are replenished throughout the lake. This process is called autumn overturn.
- As winter approaches, the surface water is eventually cooled below 4° C. At this point, the water no longer sinks. The water molecules begin to align themselves (form more hydrogen bonds) to solidify. As water temperatures at the surface reach 0°C, ice begins to cover the surface of the lake. During the winter, ice cover prevents wind from mixing the lake water. Again, stratification can occur. A layer of low density water colder than 4°C, but warmer than 0°C forms just under the ice. Below this water, the remainder of the lake water is usually near 4° C. At this point, a lake is said to be in winter stagnation. As spring approaches, the seasonal cycle begins again.
- This is beneficial because it keeps the water from being stagnant and losing its water quality. Makes the seasons the seasons and the creatures living within the environment who they are.
Water Quality
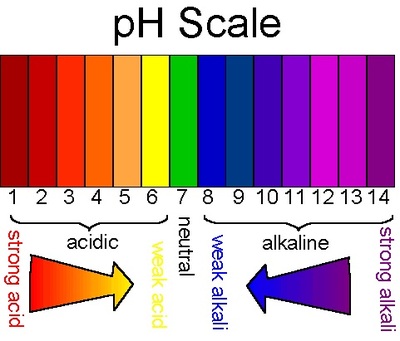
- Typical pH levels vary due to environmental influences, particularly alkalinity. The alkalinity of water varies due to the presence of dissolved salts and carbonates, as well as the mineral composition of the surrounding soil. In general, the higher the alkalinity, the higher the pH; the lower the alkalinity, the lower the pH ⁶. The recommended pH range for most fish is between 6.0 and 9.0 with a minimum alkalinity of 20 mg/L, with ideal CaCO3 levels between 75 and 200 mg/L ²⁰.
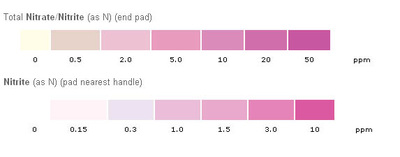
- Nitrogen-containing compounds act as nutrients in streams and rivers. Nitrate reactions [NO3-] in fresh water can cause oxygen depletion. Thus, aquatic organisms depending on the supply of oxygen in the stream will die. The major routes of entry of nitrogen into bodies of water are municipal and industrial wastewater, septic tanks, feed lot discharges, animal wastes (including birds and fish) and discharges from car exhausts. Bacteria in water quickly convert nitrites [NO2-] to nitrates [NO3-]. Nitrites can produce a serious condition in fish called "brown blood disease." Nitrites also react directly with hemoglobin in human blood and other warm-blooded animals to produce methemoglobin. Methemoglobin destroys the ability of red blood cells to transport oxygen. This condition is especially serious in babies under three months of age. It causes a condition known as methemoglobinemia or "blue baby" disease. Water with nitrite levels exceeding 1.0 mg/l should not be used for feeding babies. Nitrite/nitrogen levels below 90 mg/l and nitrate levels below 0.5 mg/l seem to have no effect on warm water fish.
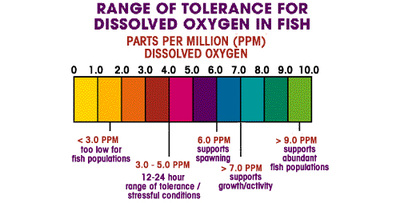
- Dissolved oxygen is necessary to many forms of life including fish, invertebrates, bacteria and plants. These organisms use oxygen in respiration, similar to organisms on land. Fish and crustaceans obtain oxygen for respiration through their gills, while plant life and phytoplankton require dissolved oxygen for respiration when there is no light for photosynthesis 4. The amount of dissolved oxygen needed varies from creature to creature. Bottom feeders, crabs, oysters and worms need minimal amounts of oxygen (1-6 mg/L), while shallow water fish need higher levels (4-15 mg/L)⁵.
Sources:
http://en.wikipedia.org/wiki/River_ecosystem
http://laurenkfreshwater.blogspot.com/2013/04/lentic-system.html
http://en.wikipedia.org/wiki/Lake_ecosystem
http://wgbis.ces.iisc.ernet.in/biodiversity/pubs/ETR/ETR25/ecosystem.htm
http://snr.unl.edu/aboutus/who/people/faculty-member.asp?pid=109
http://delaneynfreshwater.blogspot.com/2013/04/oligotrophic-mesotrophic-and-eutrophic.html
http://commons.wikimedia.org/wiki/File:Eutrophic_pond.JPG
http://jhamann.hubpages.com/hub/The-Biodiversity-of-Organisms-Found-in-Curtz-Lake-Littoral-Photic-and-Littoral-Aphotic-Zones
http://iws.collin.edu/biopage/faculty/mcculloch/2406/Notes/Aquatic%20Ecosystems/Lentic/Lentic.htm
http://www.britannica.com/EBchecked/topic/288440/inland-water-ecosystem/70743/Population-and-community-development-and-structure
http://www.anselm.edu/homepage/bpenney/teaching/BI320/CoursePages/Definitions/tropholyticzone.html
http://www.anselm.edu/homepage/bpenney/teaching/BI320/CoursePages/Definitions/Trophogeniczone.html
http://faculty.gvsu.edu/videticp/stratification.htm
https://www.truthinaging.com/review/glycolic-acid-and-ph-levels-getting-the-balance-right
https://www.discovertesting.com/articles/7,1.html
http://rahfish.com/pros-view/fishing-the-conditions/importance-of-dissolved-oxygen/
http://www.fondriest.com/environmental-measurements/parameters/water-quality/ph/
http://www.fondriest.com/environmental-measurements/parameters/water-quality/dissolved-oxygen/
http://en.wikipedia.org/wiki/River_ecosystem
http://laurenkfreshwater.blogspot.com/2013/04/lentic-system.html
http://en.wikipedia.org/wiki/Lake_ecosystem
http://wgbis.ces.iisc.ernet.in/biodiversity/pubs/ETR/ETR25/ecosystem.htm
http://snr.unl.edu/aboutus/who/people/faculty-member.asp?pid=109
http://delaneynfreshwater.blogspot.com/2013/04/oligotrophic-mesotrophic-and-eutrophic.html
http://commons.wikimedia.org/wiki/File:Eutrophic_pond.JPG
http://jhamann.hubpages.com/hub/The-Biodiversity-of-Organisms-Found-in-Curtz-Lake-Littoral-Photic-and-Littoral-Aphotic-Zones
http://iws.collin.edu/biopage/faculty/mcculloch/2406/Notes/Aquatic%20Ecosystems/Lentic/Lentic.htm
http://www.britannica.com/EBchecked/topic/288440/inland-water-ecosystem/70743/Population-and-community-development-and-structure
http://www.anselm.edu/homepage/bpenney/teaching/BI320/CoursePages/Definitions/tropholyticzone.html
http://www.anselm.edu/homepage/bpenney/teaching/BI320/CoursePages/Definitions/Trophogeniczone.html
http://faculty.gvsu.edu/videticp/stratification.htm
https://www.truthinaging.com/review/glycolic-acid-and-ph-levels-getting-the-balance-right
https://www.discovertesting.com/articles/7,1.html
http://rahfish.com/pros-view/fishing-the-conditions/importance-of-dissolved-oxygen/
http://www.fondriest.com/environmental-measurements/parameters/water-quality/ph/
http://www.fondriest.com/environmental-measurements/parameters/water-quality/dissolved-oxygen/